Lewis Structure
|
Color Key:
Orange= Hydrogen atom Blue= Carbon atom Purple= Oxygen atom Molecular modelColor Key:
Blue= Carbon atom Green= Oxygen atom Pink= Hydrogen atom |
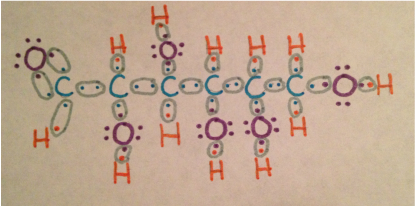
The first drawing shows the bonding between the valence electrons of the atoms in the molecule. Linear glucose has a molecular formula of C6H12O6. The bonds between all bu tone of the atoms are single bonds while one Carbon-Hydrogen bond on the end is a double bond. There are four different types of bonds in the linear glucose molecule: Carbon-Carbon, Carbon-Hydrogen, Carbon-Oxygen, and Oxygen-Hydrogen. The Carbon-Carbon bond is completely covalent because they are the same atom making the electronegativity 0. The Carbon-Hydrogen bond is very covalent because the electronegativity is 0.3. The bond between Carbon and Oxygen has an electronegativity of 1 making it moderately covalent. Lastly, the Oxygen-Hydrogen bond is weakly covalent because its electronegativity is 1.3. All of these bonds are polar. Each of the bonds are not equally shared causing a slight charge on the ends. Polar molecules are also hydrophilic, which means they are soluble in water. Since this is the linear form of glucose there is no specific VSEPR type.
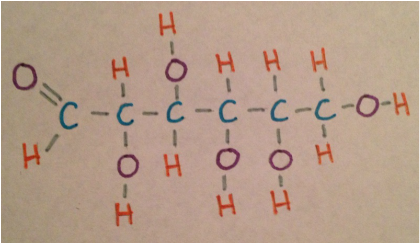
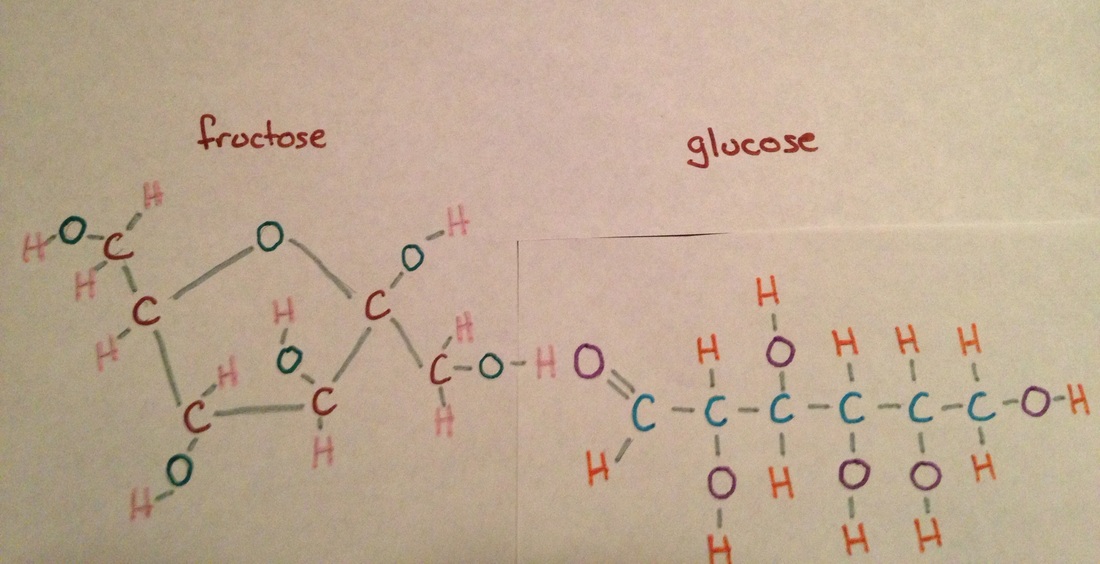
The second drawing is the Lewis structure of linear glucose which shows the completed bonds of each of the atoms. The last drawing shows linear glucose bonding to fructose. The two together form sucrose (table sugar) in the reaction: C6H12O6 + C6H12O6 ---> C12H22O11 + H2O In the bond the hydrogen of the fructose has bonded with the oxygen of the glucose to create a hydrogen bond that is a dipole-dipole because they are both polar molecules. The last picture shows the physical model made of clay and paint to show the three dimensional version of linear glucose. The clay balls represent the different atoms in linear glucose and the toothpicks represent the bonds between the atoms. |